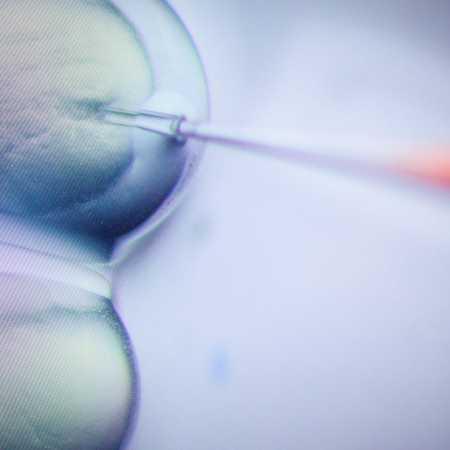
The worlds of medicine and technology became even more closely entwined this week, with health regulators in both the U.S. approving CRISPR technology in a medical context for the first time. The two treatments, Casgevy and Lyfgenia, are designed to treat sickle cell disease for patients beginning at the age of 12. This follows a similar decision from the U.K.’s Medical Healthcare products Regulatory Agency, which approved Casgevy last month to treat both sickle-cell disease and transfusion-dependent β-thalassemia.
CRISPR technology has reached a number of milestones in recent years, including Emmanuelle Charpentier and Jennifer A. Doudna receiving the 2020 Nobel Prize in Chemistry for their work developing the technology.
In announcing the agency’s approval, Nicole Verdun of the FDA’s Office of Therapeutic Products addressed precisely why this was such a milestone. “Gene therapy holds the promise of delivering more targeted and effective treatments, especially for individuals with rare diseases where the current treatment options are limited,” Dr. Verdun said in a statement.
Writing at Live Science, Emily Cooke explained in more detail just what this gene therapy does. The process causes patients’ bodies to continue producing fetal hemoglobin as opposed to the version of hemoglobin found in adults, which is at the root of both sickle-cell disease and transfusion-dependent β-thalassemia.
This Tomato Is the First CRISPR-Edited Food to Go on Sale
They’re just hitting Japan now, but expect more gene-edited food to followWhile the process could have significant benefits for people affected by these conditions, the treatment also includes a long recovery period. Live Science’s reporting noted the MHRA’s finding that patients might require “at least a month in a hospital facility” to adjust to the changes in their bodies. These regulatory approvals are a big step forward — but they’re only one part of a longer recovery process.
The Charge will help you move better, think clearer and stay in the game longer. Subscribe to our wellness newsletter today.