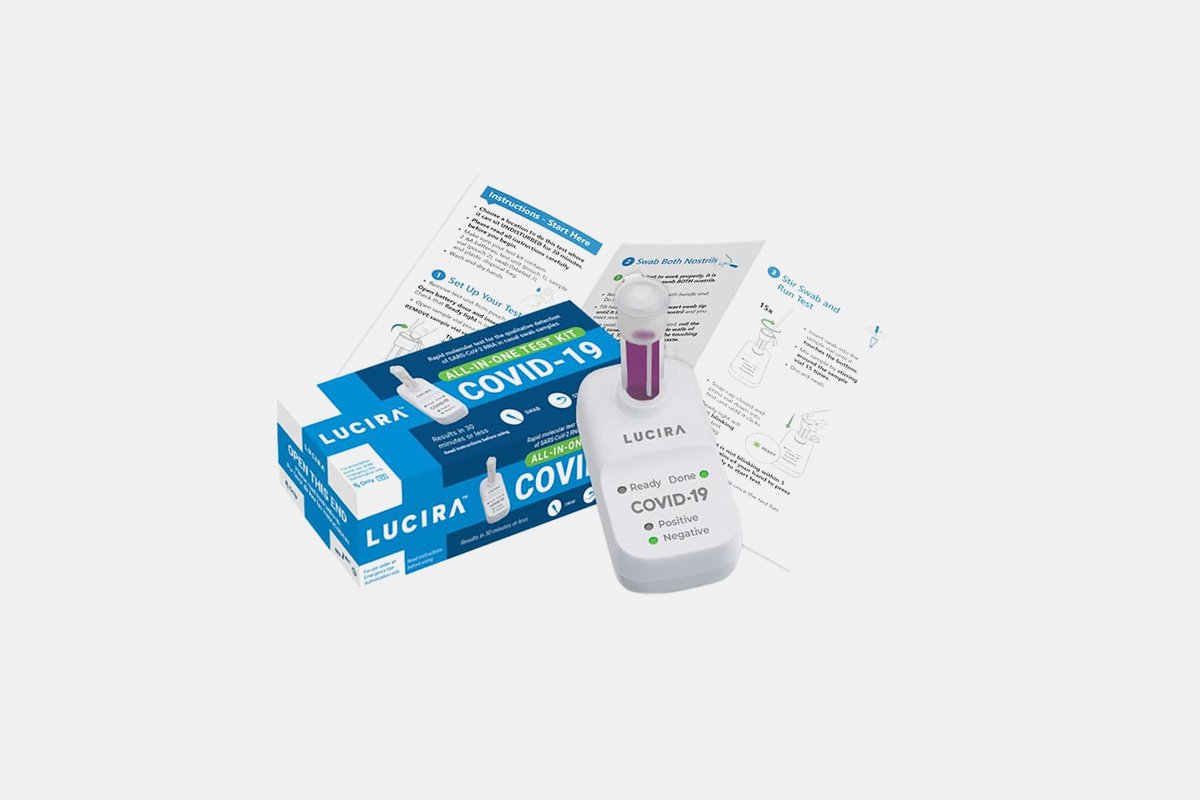
California biotechnology lab Lucira Health has successfully developed the nation’s first FDA-approved, at-home coronavirus rapid test. Called the “All-In-One” test kit, it includes a vial of solution and a portable, battery-operated device. Once nasal specimen is mixed with the solution, the device takes anywhere from 11 minutes to a half hour to provide a result. A light will indicate whether the test came back negative or positive.
For now, Lucira is only making these kits available for two specific sites: Northern California’s Sutter Health and South Florida’s Cleveland Clinic. National rollout won’t happen until spring of next year. While the test is highly accurate (agreeing with results from traditional clinic testing 94-98% of the time), Lucira needs time to scale up its manufacturing capabilities.
It’s unclear, right now at least, exactly what sort of impact these test kits could have. After all, pharmaceutical companies Pfizer and Moderna have also tapped next spring for the rollout of their coronavirus vaccines, assuming they manage to secure emergency use authorization from the FDA in the coming weeks.
Still, it will take much of next year to vaccinate the public — assuming that most Americans are even willing to get vaccinated. (Yesterday’s poll showed that percentage crawl up to 58% of the public, which is still astonishingly low.) Plus, let’s remember, America is currently reporting well over 100,000 cases a day. There is little guarantee that this virus will be gone by the end of March.
These tests, which Lucira says will cost just $50, could provide an alternative to visiting the hospital or waiting on a long line outside a clinic. Unvaccinated people, those concerned they have the virus, and any who need a test to, say, board a plane, would be able to obtain the All-In-One kit with a doctor’s prescription, and have peace of mind — or the knowledge that they need to start quarantining — in a matter of minutes.
The Charge will help you move better, think clearer and stay in the game longer. Subscribe to our wellness newsletter today.