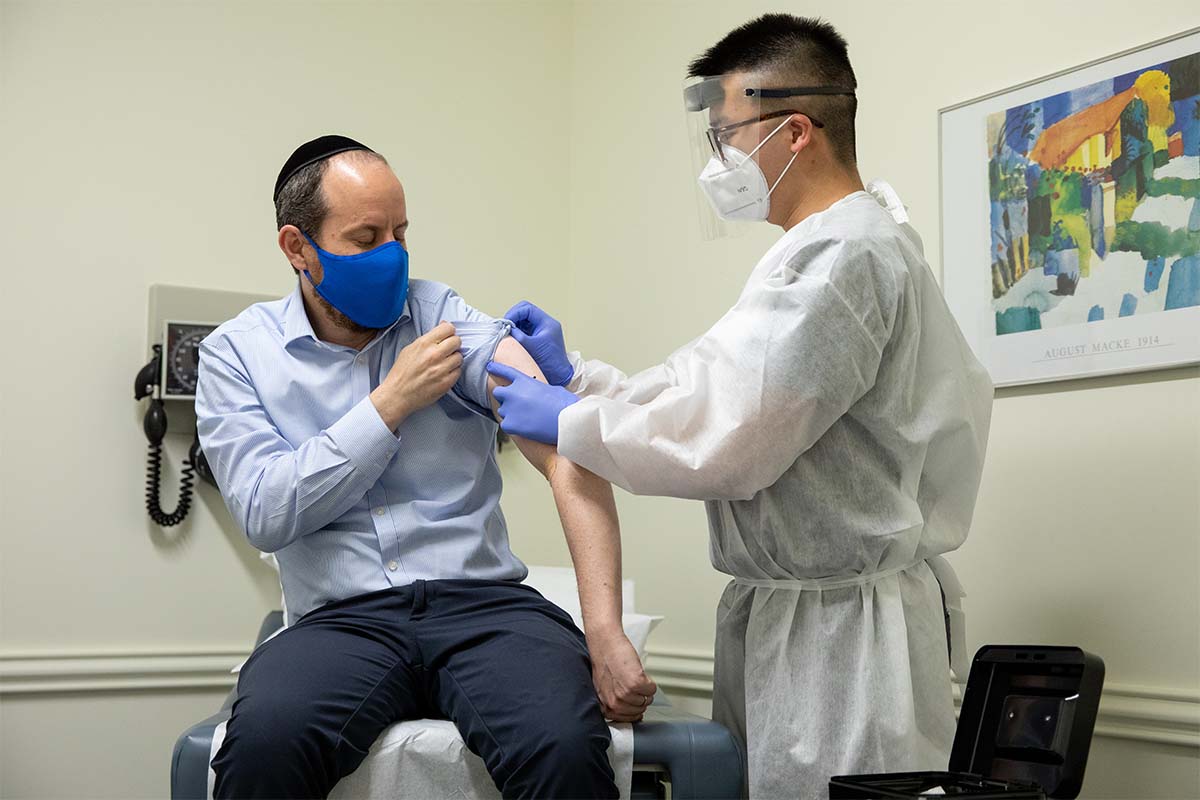
Given that we’re on second and third outbreaks already, it looks like we’re stuck in semi-quarantine as the coronavirus continues to spread.
Our next big hope? A vaccine. And while Dr. Anthony Fauci was “cautiously” optimistic we’ll have one by year’s end or early 2021, we’re in a holding pattern right now until the various scientific trials are finished.
But that means human trials. And yes, you might be able to get into one early.
As Arlene Weintraub of Next Avenue reports, there are opportunities for everyday people to participate in the COVID-19 Prevention Network (COVPN), an initiative of the National Institutes of Health’s National Institute of Allergy and Infectious Diseases.
If you are interested in volunteering for a COVID-19 clinical trial, however, there are a few things you need to know. As Weintraub notes, each of the participating companies will need about 30,000 volunteers for their respective attempts.
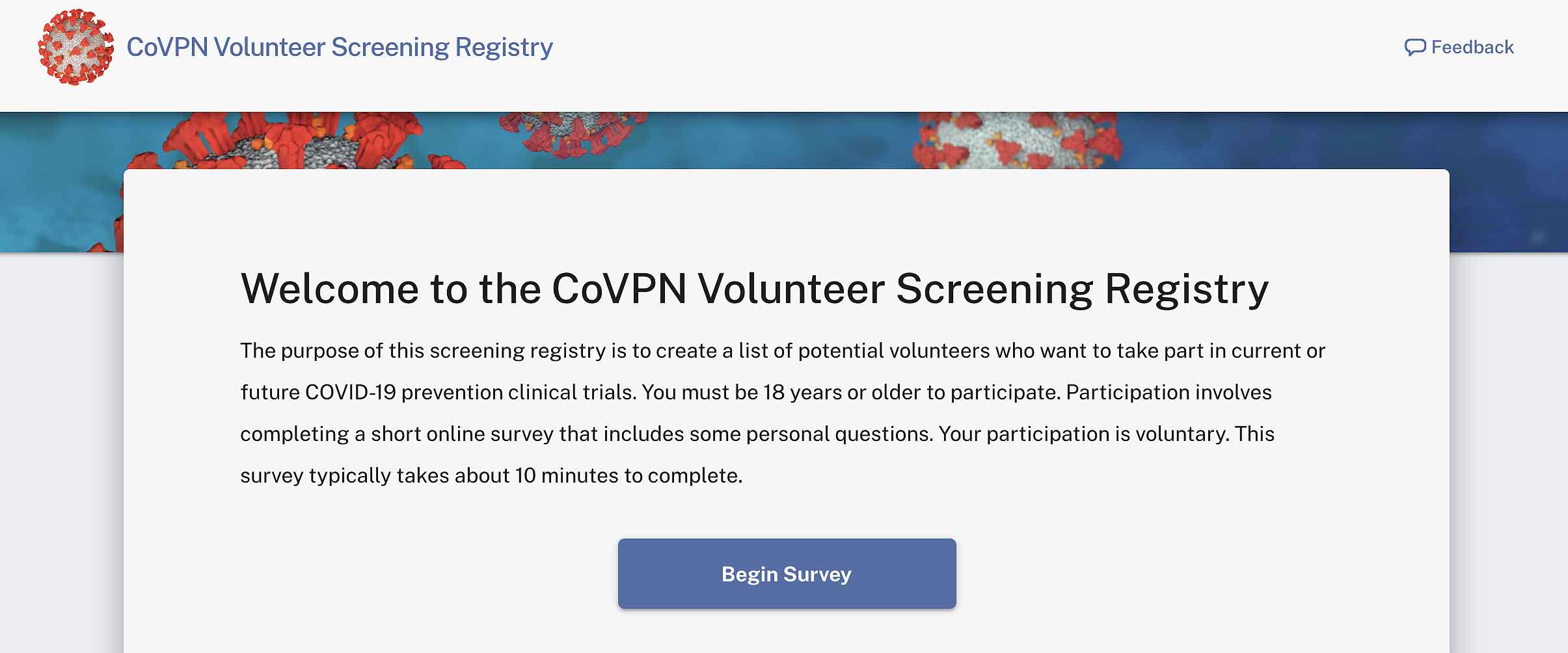
Each company working on a vaccine has their own requirements. “People with pre-existing conditions are a priority population, as are adults over age 65,” says Michele Andrasik, a scientist in the vaccine and infectious disease unit at the Fred Hutchinson Cancer Research Center. So are more at-risk populations like African Americans, Native Americans and Latinos, as well as people who work at health care facilities or in open work spaces (e.g. meatpacking plants).
And while you may or may not fit the above criteria, your current medications might disqualify you from some trials. That said, the large number of volunteers needed means you could be put on a list of potential participants.
A few other things to note: There’s a 50% chance you’ll receive a placebo. And you might experience side effects. And you still might actually get sick with the coronavirus anyway. “We’re not exposing people to COVID-19 or giving it to them, but we do have to find people who have a high risk of getting it,” explains Dr. Richard Novak, professor of medicine and chief of the infectious disease unit at the University of Illinois and one of the clinical trial investigators for Moderna.
As well, there will be plenty of phone, email and in-person follow ups — which could last up to two years — and probably only modest compensation at best.
That said, if the vaccine doesn’t appear to be working or there are any complications, the trials can be suspended quickly.
Subscribe here for our free daily newsletter.
Whether you’re looking to get into shape, or just get out of a funk, The Charge has got you covered. Sign up for our new wellness newsletter today.